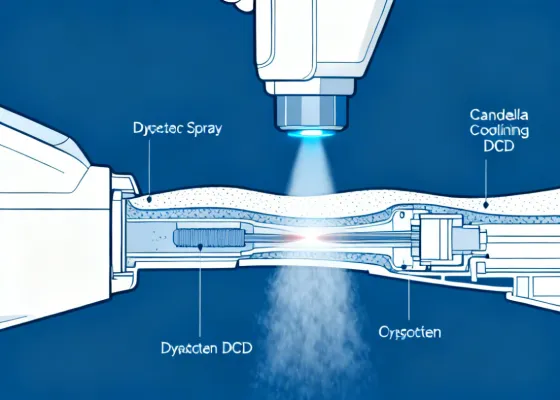
Candela Laser Technology & Innovation
Behind every Candela system is decades of laser research, proprietary engineering, and a relentless focus on what works in the real world of clinical practice. Here's a closer look at the technology that makes it possible.